The Indian market for Glucagon-Like Peptide-1 (GLP-1) therapies is currently at an inflection point and is poised for strong expansion. The current market size for anti-obesity and diabetes GLP-1 drugs in India, as of 2025, is estimated to be between ₹1,000-1,200 crore. Notably, the global market size reached about US $72 billion (₹6.8 lakh crore) in FY25.
The 5X growth potential to ₹5,000 crore
According to CareEdge Ratings, this market is expected to grow nearly 5X to ₹5,000 crore by 2030. Despite this, drug penetration remains negligible currently, but it is expected to rise to around 1% by then due to increased marketing efforts, price rationalization, and a growing patient pool.
From a therapeutic standpoint, an estimated 60% to 70% of the demand will be driven by type-2 diabetes treatments, while the remainder will be for weight-loss applications. The patent expiry of Semaglutide in India on 20 March 2026 is the major catalyst for the Indian GLP-1 market.
Catalyst 2026: The patent cliff
Upon patent expiry, drug prices are expected to drop by 40% to 50% in FY27, with an additional 10% to 30% reduction in FY28 due to heightened competition. A few Indian companies have already secured regulatory approvals for generic versions of Semaglutide, and an additional 10 to 20 companies are actively developing their own formulations.
Similarly, this article discusses three early-mover companies with different pricing strategies: one offers premium pricing, another focuses on mass-market affordability, and the third holds distribution rights to the Novo Nordisk GLP-1 drug.
#1 Sun Pharmaceuticals: The affordability disrupter
Sun Pharmaceutical is the world’s leading specialty generics pharmaceutical company and India’s largest pharmaceutical company, holding an 8.4% share of the domestic market. Globally, it ranks as the 13th largest generics manufacturer in the US. It operates in over 100 countries.
The company operates diverse businesses across different geographies and product categories. As of FY25, India formulations accounted for 33% of sales, the US (31%), emerging markets (18%), and the rest of the world (14%).
Sun Pharma launched its generic semaglutide injections in the Indian market on 21 March 2026. The company positioned itself to be in the market on day one, adding dedicated field force personnel to support the launch.
Scaling affordability with “noveltreat”
These products are manufactured in Europe using a pen device and sold in India under two different brands: Noveltreat and Sematrinity. A core focus of this launch is improving affordability and expanding access to therapies that are highly effective for treating obesity and type 2 diabetes.
Disrupting the mass market: Sun Pharma’s ₹750 entry
Accordingly, Sun Pharma has priced these drugs significantly lower than the brands mentioned, with weekly therapy costs ranging from ₹900 to ₹2,000 for Noveltreat and from ₹750 to ₹1,300 for Sematrinity.
Beyond Generics: The GL0034 innovation pipeline
In addition to generic offerings, Sun Pharma is building its global innovative medicines portfolio by developing its own novel GLP-1 receptor agonist, GL0034. The drug is currently in global Phase II clinical trials for type 2 diabetes.
It expects to publish Phase-2A data for both diabetes and NASH (nonalcoholic steatohepatitis) at scientific conferences shortly. The ongoing Phase-2b study is expected to be completed within 12-18 months. From a financial perspective, Sun Pharma reported double-digit growth in sales and profit.
9M Financials: Sustaining 30%+ EBITDA margins
Sales in 9MFY26 increased 11% year-over-year to ₹43,660 crore. EBITDA (earnings before interest, taxes, depreciation, and amortization) increased 19% to ₹13,777 crore, with a margin of 31.4%. Net profit increased 6% to ₹9,651 crore.
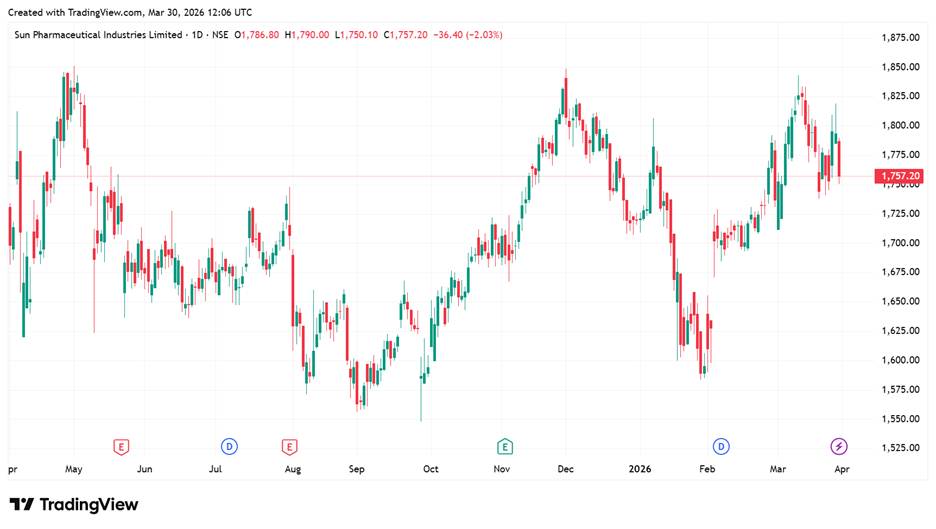
Sun Pharma Share Price
#2 Dr. Reddy’s Laboratories: In-House tech and global scaling
Dr. Reddy’s Laboratories is a leading therapeutic company offering products in gastrointestinal, cardiovascular, diabetes, oncology, pain management, and dermatology. It has a presence in the USA, India, Russia, CIS countries, China, Brazil, and Europe.
Obeda: India’s First Approved Premium Generic
Dr. Reddy’s has also made a Day-1 entry into the GLP-1 receptor agonist therapy space with the launch of its injectable generic semaglutide, marketed under the brand name Obeda. It aims to build a full-breadth GLP-1 portfolio targeting multiple metabolic disorders, such as diabetes and obesity.
The premium pricing strategy
Obeda is India’s first DCGI-approved generic semaglutide injection. It is available in a pre-filled disposable pen for subcutaneous, once-a-week use. Each pen delivers a minimum of 4 weekly doses, and pricing is premium at ₹4,200 per month for both strengths. Dr. Reddy’s developed Obeda completely in-house.
Vertical Integration: A 1.2 Crore Pen Capacity
The company currently has a capacity of 1.2 crore pens. While they are initially utilizing their partner Stelis for the fill-and-finish process, Dr. Reddy’s plans to expand capacity using their own internal facilities in the future. Going ahead, Dr. Reddy also plans to introduce oral semaglutide.
Eyeing Global Launch in Over 80 Countries
Dr. Reddy’s aims to launch its generic semaglutide in over 80 countries. Upcoming launches are targeted for Brazil and Turkey around July 2026, and are actively pursuing regulatory approval in North America. It expects a potential launch in Canada between February and May 2026, assuming Health Canada clears their application without requiring a plant inspection.
From a financial perspective, Dr. Reddy’s Laboratories reported single-digit growth in sales and a decline in profit. Revenue in Q3FY26 increased 4.4% year-over-year to ₹8,727 crore. EBITDA (earnings before interest, taxes, depreciation, and amortization) decreased 11% to ₹2,049 crore, with a margin of 23.5%. Net profit decreased 14% to ₹1,210 crore.
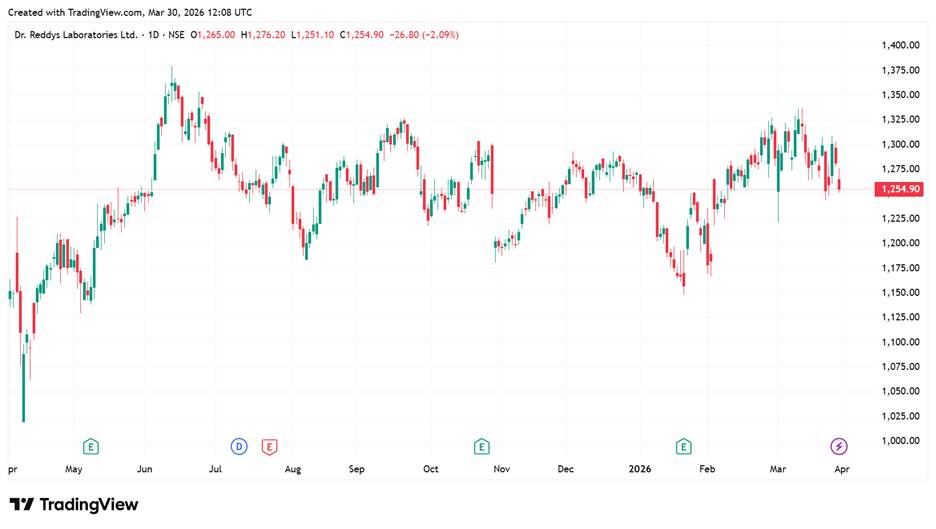
Dr. Reddy’s Share Price
#3 Emcure Pharmaceuticals: The Exclusive Innovator Partnership
Emcure Pharmaceuticals has a strong presence in Gynecology and Cardiovascular. Its business is currently split roughly 50-50 between chronic and acute therapies. Emcure ranks #2 in Gynaecology in India. Domestic accounts for 45% of trailing twelve months revenue, while export markets make up the remaining 55%.
Poviztra: Leveraging the Novo Nordisk Legacy
In late 2025, Emcure secured an exclusive partnership with Novo Nordisk, beating out at least 8 of 9 competitors. Through this agreement, Emcure received exclusive rights to distribute and commercialize Novo Nordisk’s GLP-1 innovator drug in India.
Emcure launched this product at the end of December 2025 under the brand name Poviztra, priced at ₹8,790 per month. It is semaglutide injection specifically targeted for weight loss and the treatment of obesity, and its related comorbidities. Management noted that obesity is associated with roughly 230 comorbidities, making this a highly significant therapeutic area.
Cardiovascular Footprint as a Growth Multiplier
Management anticipates strong generic competition but remains highly confident in Poviztra’s clinical advantages. Compared with other treatments like Tirzepatide, management highlights that emerging data show Semaglutide is as effective as, if not better than, Tirzepatide for weight loss and is far better.
To maintain an edge over generic peers, Emcure plans to rely on the extensive clinical data of the innovator drug, the superior convenience of the injection device, and pricing flexibility. To maximize the drug’s reach, Emcure is utilizing a highly complementary strategy with Novo Nordisk.
Emcure is leveraging its massive sales footprint in the Cardiovascular segment to promote the drug, backed by strong cardiac data. Two of Emcure’s largest divisions are handling the promotion and education of the medical fraternity, Pan-India.
R&D Pivot: Transitioning to In-House GLP-1 Delivery
In addition to the in-licensed Poviztra, Emcure is investing in in-house research in the GLP-1 category. Its mid- to long-term pipeline includes the development of a novel semaglutide anti-diabetes drug delivery system. Emcure anticipates that semaglutide will eventually become a highly meaningful growth driver for their business in international and emerging markets.
From a financial perspective, Emcure Pharmaceuticals reported double-digit growth in sales and a strong increase in profit. Revenue in Q3FY26 increased 20.4% year-over-year to ₹2,363 crore. EBITDA increased 27% to ₹460 crore, with 19.5% margin. Net profit grew 48% to ₹231 crore.
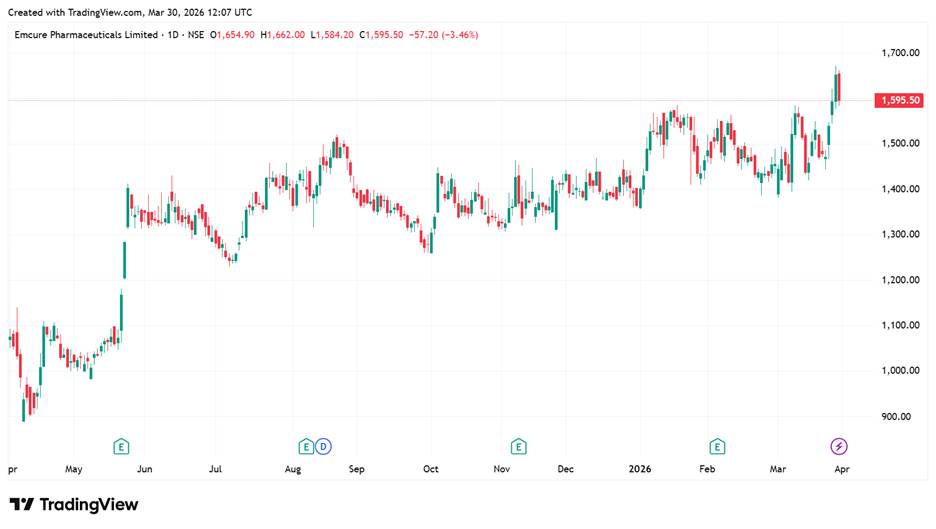
Emcure Share Price
Valuation Check: Are These GLP-1 Plays Overpriced?
Return ratios, including both return on capital employed (ROCE) and return on equity (ROE), are strong across all three companies. Valuation-wise, Sun Pharma and Emcure are trading almost in line with historical multiples, but at a premium to industry valuation. On the other hand, Dr. Reddy’s is trading in line with historical multiples, but at a discount to the industry.
| Peer Comparison (X) | ||||
| Company | P/E | 5Y Median P/E | ROCE (%) | ROE (%) |
| Sun Pharma | 35.0 | 32.0 | 20.2 | 16.9 |
| Dr. Reddy’s | 18.8 | 20.0 | 22.7 | 18.0 |
| Emcure | 33.4 | 35.4 (~2Y) | 20.7 | 18.5 |
| Industry Median | 26.5 | – | 15.1 | 13.1 |
The GLP-1 opportunity is shifting from a niche to a scalable market, driven by patent expiries, price cuts, and rising awareness. Early movers with differentiated strategies across pricing, innovation, and distribution are best positioned to capture this expanding therapeutic and commercial opportunity. Meanwhile, add them to your watchlist and stay tuned.
Disclaimer
Note: Throughout this article, we have relied on data from http://www.Screener.in and the company’s investor presentation. Only in cases where the data were unavailable have we used an alternative, widely accepted, and widely used source of information.
The purpose of this article is only to share interesting charts, data points, and thought-provoking opinions. It is NOT a recommendation. If you wish to consider an investment, you are strongly advised to consult your advisor. This article is strictly for educational purposes only.
About the Author: Madhvendra has been deeply immersed in the equity markets for over seven years, combining his passion for investing with his expertise in financial writing. With a knack for simplifying complex concepts, he enjoys sharing his honest perspectives on startups, listed Indian companies, and macroeconomic trends.
A dedicated reader and storyteller, Madhvendra thrives on uncovering insights that inspire his audience to deepen their understanding of the financial world.
Disclosure: The writer and his dependents do not hold the stocks discussed in this article.
The website managers, their employees (s), and contributors/writers/authors of articles have or may have an outstanding buy or sell position or holding in the securities, options on securities, or other related investments of issuers and/or companies discussed therein. The content of the articles and the interpretation of the data are solely the personal views of the contributors/writers/authors. Investors must make their own investment decisions based on their specific objectives, resources, and only after consulting such independent advisors as may be necessary.